Harnessing this energy and channeling it for the right use is a challenging task.Ītomic bombs are nuclear weapons that employ massive energy from the fission reaction to produce explosions. Apart from this, recent technological advancements have enabled the application of nuclear energy in industries such as extraction of oil, desalination of water, hydrogen production, etc. We have so far understood that a nuclear fission reaction produces a huge amount of energy. Read More:- Mass Energy and Nuclear Binding Energy However, an uncontrolled fission reaction can cause extensive damage to life and property, what we commonly know as an atomic bomb explosion. When this reaction is carried out in a controlled environment like that of a nuclear reactor, the energy produced can be utilized to produce power on a commercial scale. This made way for the generation of new reactor models and gave birth to the Nuclear Power industry. Fast neutrons were then used to bombard the Uranium 236 nucleus and it was discovered that the reaction produced energy exceeding 1 MeV and it was a self-sustaining chain reaction.Įnrico Fermi and his associates, in the year 1942 carried out this nuclear reaction in a controlled environment, inside a nuclear reactor. The most abundantly available Uranium-236 isotope was later understood to be playing a key role in this reaction. Later, these transuranic elements were investigated and found to be radioisotopes of Uranium namely, Barium and Lanthanum, and some other elements in the periodic table. Following this, physicists Otto Hahn and Strassman, in the year 1939, performed the famous nuclear experiment wherein they bombarded an isotope of Uranium with slow neutrons to produce transuranic elements and a very high amount of energy.

Three types of moderators are used at the MIT reactor: (1) ordinary or "light" water that is also used to cool the reactor core, (2) deuterated or heavy water (D 20), and (3) high-purity graphite, both of which are excellent at slowing neutrons without absorbing them.It all started in the year 1932 when the neutron, a constituent subatomic particle, was discovered by James Chadwick in England. Since U-235 nuclei do not readily absorb the high energy neutrons that are emitted during fission, it is necessary to slow the neutrons down with a "moderator". In the MIT reactor, one other group of components is essential to the maintaining and controlling a chain reaction. As fewer and fewer neutrons are absorbed, more and more neutrons are available to cause the splitting of uranium nuclei, until finally enough neutrons are available to sustain a chain reaction. To put the reactor into operation, the control blades are raised very slowly. When the control blades are fully inserted, they absorb so many neutrons from the uranium that there are not enough to allow a chain reaction to continue. Boron has the property of absorbing neutrons without re-emitting any. The rate of fissions in the uranium nuclei in the MIT reactor is controlled chiefly by six control blades of boron-stainless steel which are inserted vertically alongside the fuel elements. When it is in operation, the central active core contains a huge number of neutrons traveling in every direction at very high speeds. The MIT Research Reactor is used primarily for the production of neutrons. Hence, the possibility exists for creating a chain reaction.


Each time a U-235 nucleus splits, it releases two or three neutrons.
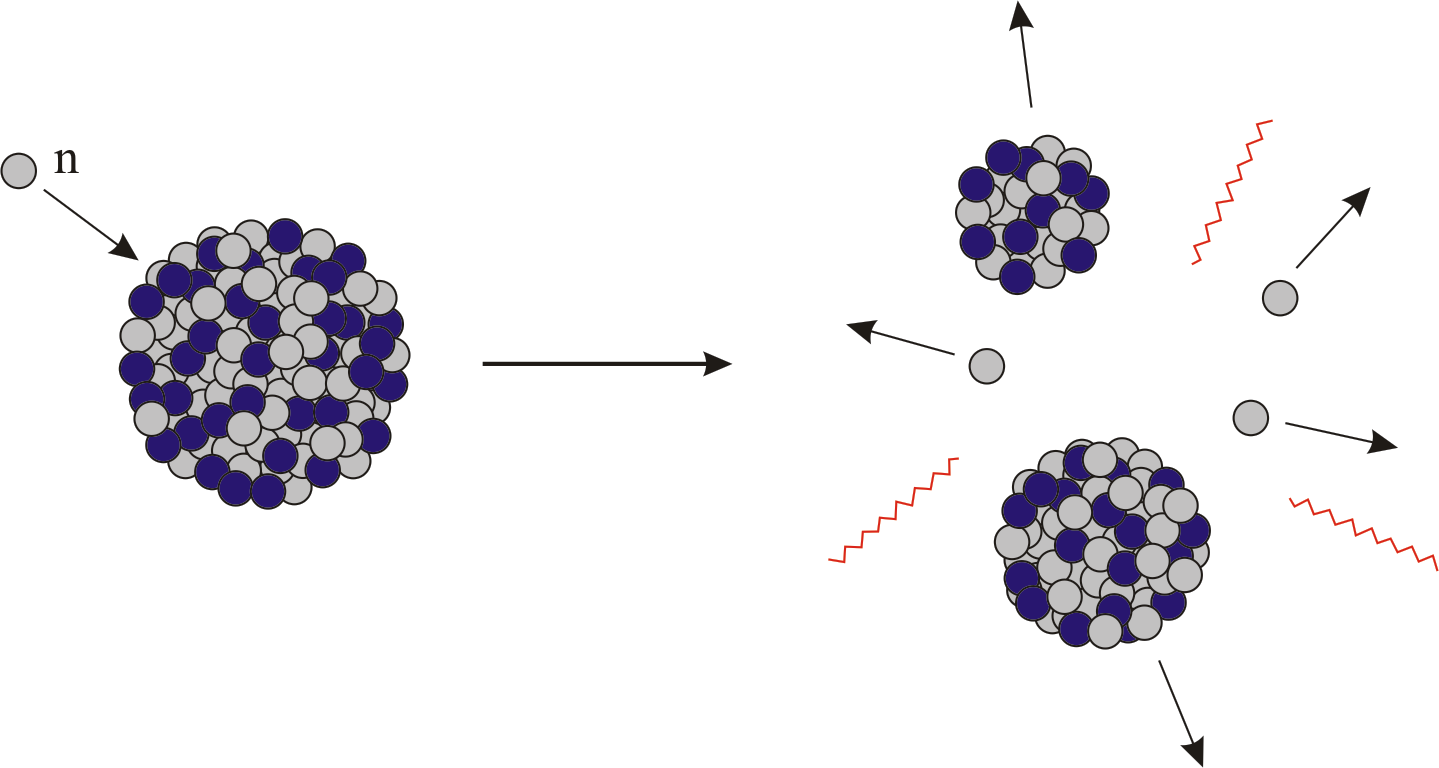
This process is known as fission (see diagram below). When a U-235 nucleus absorbs an extra neutron, it quickly breaks into two parts. The arrangement of particles within uranium-235 is somewhat unstable and the nucleus can disintegrate if it is excited by an outside source. In the nucleus of each atom of uranium-235 (U-235) are 92 protons and 143 neutrons, for a total of 235.
